Mail-Back Disposal: Why Prepaid Envelopes Won’t Solve America’s Controlled Substance Waste Problem
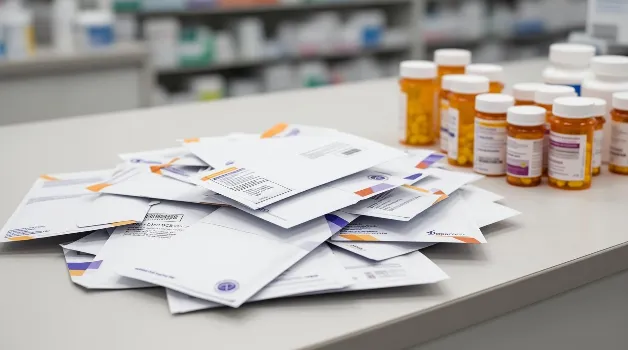
On March 31, 2025, the FDA’s Opioid Analgesic REMS mandate took effect, requiring every manufacturer of extended-release and long-acting opioid analgesics to provide prepaid, preaddressed mail-back envelopes with each dispensed prescription. The goal is straightforward: make it easier for patients to dispose of unused opioids safely. The intention is sound. The execution faces fundamental challenges […]
The Essential Features Every LTC Pharmacy Software Must Have (Most Miss #3)

Long-term care pharmacies don’t operate like retail pharmacies. Anyone who’s worked in LTC knows that comparison falls apart fast. LTC pharmacies juggle complex medication workflows, dozens (sometimes hundreds) of facilities, high-volume cycle fills, constant census changes, and regulatory pressure that never lets up. Yet many platforms marketed as LTC pharmacy software are still retail systems underneath, dressed up with […]
Amyotrophic Lateral Sclerosis (ALS) Fund

Advancing Rapid Microbial Detection with Celsis®

Why settle for slow, traditional methods when speed and accuracy are within reach? Celsis® minimizes change and strengthens your regulatory path. Get faster results, greater capacity, support for complex materials, and digital reporting. Learn how to stay ahead of evolving regulations and lead the future with confidence in this brochure. Read More >
We’re Taking No Chances When It Comes to Compliance

Every controlled substance transaction should be secure, traceable, and protected. As compliance standards continue to evolve, pharmacies need stronger safeguards to ensure every record stands up to audits and inspections. That’s why C2 Keep has transitioned from 4-digit to 6-digit PINs to strengthen transaction security and reinforce accountability across your team. Why Stronger PINs Matter? […]
From Good to Best-in-Class: Specialty Pharmacy Benchmarks and the Data-Driven Path to Better Outcomes

Health system specialty pharmacies are uniquely positioned to deliver exceptional patient impact but as expectations rise, so does the need to prove performance with consistency, clarity, and measurable outcomes. What separates programs that are doing well from those operating at best-in-class levels? In this webinar, we’ll explore a practical Good / Better / Best framework […]
On the Road: March Highlights

We wrapped a strong showing at: From packed booths to meaningful conversations, one thing was clear:There’s real demand for faster, more confident clinical decision-making. And FebriDx is right at the center of it. Follow along for more field moments:LinkedIn – PHASE Scientific AmericasLinkedIn – FebriDx Next stops: come see usWe’re not slowing down. Catch us […]
The Essential Guide to Documentation and Certification for Sterilizing-Grade Filters

Microbial contamination is one of the most serious threats in drug manufacturing. As a result, preventing it starts long before a product ever reaches the patient. That risk is why sterilizing-grade filtration is crucial in industries like pharma and biotech. In drug manufacturing, 0.2µm sterilizing-grade filters are used to remove potentially harmful bacterial contaminants and […]
Dexcom G6 is Being Discontinued

SRx’s Next Generation Suppository Molds

Are you tired of struggling with traditional PVC suppository shells? Is messy spillage and inefficient workflow slowing down your pharmacy? Say goodbye to those frustrations and launch your suppository compounding into the future with RocketMolds™ Suppository Compounding Molds! Why Choose RocketMolds™? Sizes: CLICK HERE to order online! See also:RocketMolds™ Suppository Compounding Mold HolderAvailable with or without LED Backlight Shipping […]
