How revised chapters of USP regulations impact pharmaceutical production to increase patient safety
This December, the United States Pharmacopeia (USP) updates for General Chapters (GC) <795> and <797> will be official and mandate major changes to compounders’ processes. USP <795> Pharmaceutical Compounding – Nonsterile Preparations and <797> Pharmaceutical Compounding – Sterile Preparations will incorporate major chapter changes directly impacting compounding pharmacies with the overall intention of increasing quality of products to minimize patient risk and harm. If there is one thing the 2012 compounding pharmacy public health tragedy taught us, it is that regulations and proper quality control testing save lives. The under-regulated pharmacy caused 64 deaths and 753 sickened, according to the US Centers for Disease Control and Prevention, caused from a fungal meningitis outbreak through contaminated products (Ref. 3).
Changes to these chapters reflect advancements in science and clinical practice, clarification of sections that were frequently misconstrued, and input from more than 6,400 public comments received. (Ref.1). These chapters outline an increased requirement for quality assurance by increasing environmental monitoring schedules, enhancing validated processes for detection and identification of microbial contamination, and monitoring personnel and raw materials proving and maintaining quality pharmaceuticals are being manufactured.
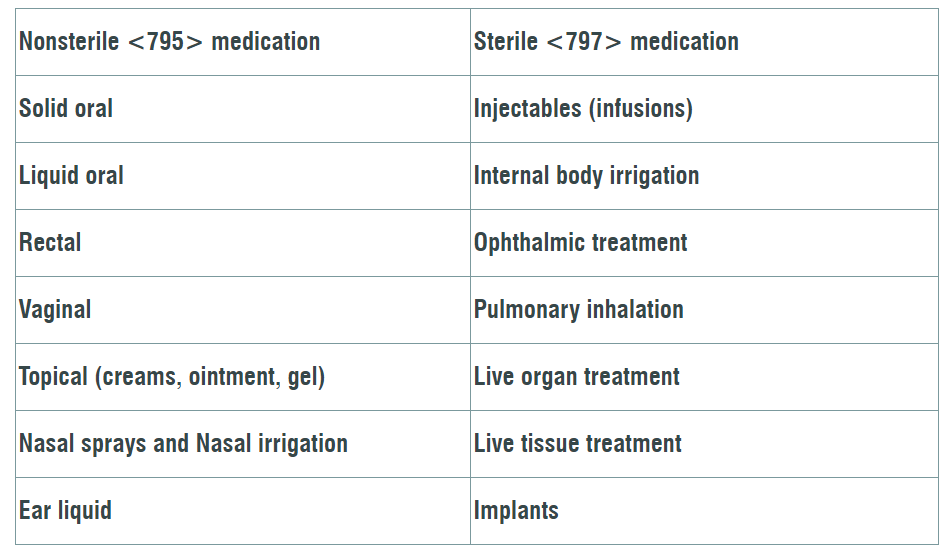
Highlighting the changes to the General Chapter <795> and <797>
Updates to <795> discussed in this article:
- Elimination of the Categories of Compounding, which formerly included simple, moderate, and complex. The categories of compounding did not impact how the standards were applied but frequently caused confusion on how to classify the preparations.
- Clarification of the scope of the chapter to include types of Compounded Nonsterile Preparations (CNSP) and to exclude specific practices including:
- Administration
- Nonsterile radiopharmaceuticals
- Reconstitution
- Repackaging
- Splitting tablets
- Inclusion of new provisions for equipment for weighing, measuring, or otherwise manipulating components that could generate airborne particles. Facilities must assess these activities to determine whether a closed system processing device is required to reduce potential exposure to personnel or contamination of the facility or CNSP. Examples of closed system processing devices include:
- Containment ventilated enclosures (CVEs)
- Biological safety cabinets (BSCs)
- Single-use containment glove bags
Updates to <797> discussed in this article:
- Revision of the microbial contamination risk levels (i.e. low-, medium-, and high-risk level) to Category 1 and Category 2 compounded sterile products (CSPs). Category 1 and Category 2 are distinguished primarily based on the conditions under which they are made, the probability for microbial growth, and the time period within which they must be used.
- Revision of the Facilities and Engineering Controls requirements to better describe the types of primary engineering controls (PECs) and their placement. New requirement to place Compounding Aseptic Isolator (CAI) and Compounding Aseptic Containment Isolator (CACI) in a cleanroom suite to prepare Category 2 CSPs, which have longer Beyond Use Dates (BUDs), to minimize the risk of contamination. Movement of materials in and out of the CAI/CACI in unclassified air carries a higher risk of contamination and thus placement of the CAI and CACI in classified areas helps mitigate the risks for CSPs with longer Category 2 BUDs.
- Update to require monthly surface sampling, whereas previously surface sampling was required “periodically” and interpreted differently among healthcare facilities. Monthly surface sampling provides a measure of control and provides additional monitoring in between semi-annual (e.g. every 6 months) viable air monitoring and certification requirements.
What is Compounding?
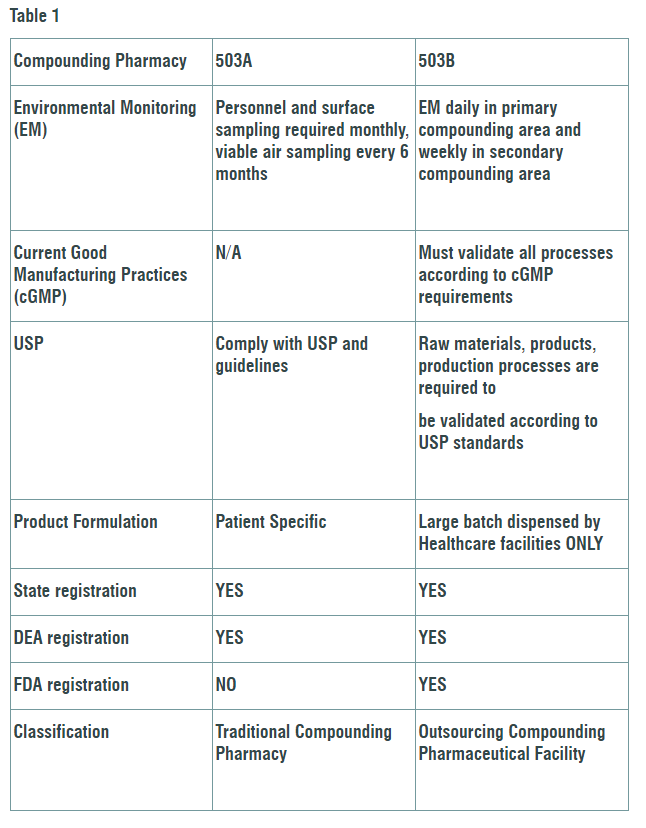
Compounding is combining and mixing 2 or more ingredients to create a unique medical compound in a dose specific to the patients’ medical needs (Ref. 2). Compounding Pharmacies are divided into two sectors. Designated 503A facilities are limited to dispensing small amounts of drugs and must follow USP and state pharmacy guidelines. Larger 503B pharmacies are allowed to produce bigger batches and must validate every process with current good manufacturing practices (cGMP). The key differences and USP required process changes that will impact both sectors differently are outlined in Table 1.
We have seen an increase in regulatory personnel and resources to perform more frequent audits in compounding pharmacies to ensure the requirements set forth are being implemented immediately. Maintaining current, compliant and regulated compounding processes in a controlled environment will ensure consistent quality pharmaceuticals, provide healthcare administrators confidence in product efficacy, and prevent risk to patients’ safety. These proposed USP chapter revisions place a greater emphasis on the requirement for conducting investigations and implementing corrective actions.
For more information about how these changes will impact the compounding pharmacy industry and how Charles River can support needed regulatory changes check out Our Compounding Pharmacy resource and services page.
References
2019. Decisions on Appeals to USP<795> and <797>
2019. New and Revised USP Compounding Standards
2015. Multistate Outbreak of Fungal Meningitis and Other Infections








