As the world tries to cope with the COVID-19 pandemic, the race for a vaccine and effective treatment against SARS-CoV-2 virus has been witnessing substantial progress with significant number of clinical trials launched across the world. However, with health officials and drug manufacturers’ indicating that we are still about 12-18 months away from successfully developing, trialling and launching a vaccine, national bodies are looking for effective containment strategies as an immediate action. A significant step towards this is the efficient testing and diagnosis of COVID-19. Testing not only allows the infected to receive adequate and appropriate care, but also helps understand the spread and take evidence-based measures to slow it down.
With most countries and laboratories ramping up testing of suspected cases, a number of test variants using different specimens have emerged. Even though the most common among them are throat swabs, saliva samples etc. for PCR (Polymerase Chain Reaction) test, some national bodies are also conducting antibody tests that require blood to be drawn and analyzed. Irrespective of the type of test, the accuracy of the test results depends on the integrity of the specimens. From their collection to the laboratory, they must be handled with the greatest care.
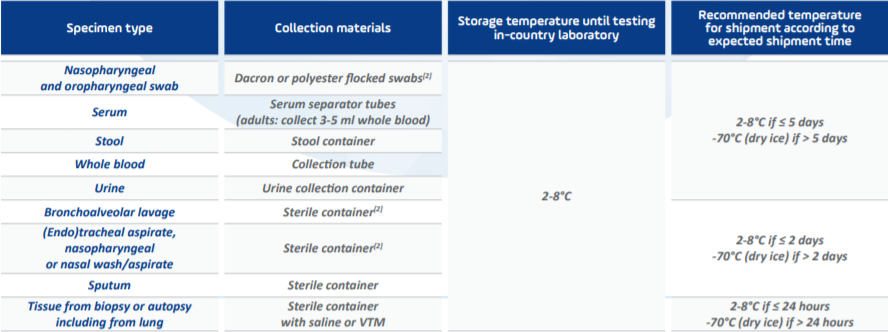
In order to ensure accurate results, it is essential to understand and implement the storage and transport conditions mandated by the regulatory bodies. According to the interim guidance provided by WHO[1], specimens for virus detection should reach the laboratory as soon as possible after collection. In addition, as per official guidelines, specimens that are delivered promptly to the laboratory can be stored and shipped at 2-8°C. However, when there is a higher likelihood of delay in the specimens reaching the laboratory, the use of viral transport medium is strongly recommended. Specimens may be frozen to -20°C or ideally to -70°C and shipped on dry ice if further\ delays are expected. More details on the specimen type and the recommended temperature range can be found in Table 1:
The regulations also mandate the use of Viral Transport Medium (VTM) for the specimens in case of delayed transportation. Even though there are country-specific regulations around the production of the VTM, some of the core reagents remain the same, with Fetal Bovine Serum (FBS) being one of the major one. FBS is best-stored frozen, between -5 to -20°C, and can be thawed at a temperature between +2 to +8°C.
This has created an unprecedented need for the maintenance of a strong and reliable cold chain. Be it the insulated boxes for transporting specimens, refrigerators for storage at 2-8°C, freezers for storage at -20°C, or ultra-low freezers for storage at -70°C and below, the integrity of the samples solely depends on the ability to maintain the intended temperature.
B Medical Systems has been supporting laboratories worldwide for the last 40 years in maintaining an effective cold chain. Through our portfolio of transport boxes, pharmacy refrigerators, laboratory refrigerators, lab freezers, blood bank refrigerators and ultra-low freezers, we are committed to offering you an unbroken cold chain and being your partner in the fight against this pandemic. For more information, visit us at https://www.bmedicalsystems.com/our-solutions/medical-refrigeration/.
B Medical Systems S.à r.l.
17, op der Hei
L - 9809 Hosingen, Luxembourg
Tel. : (+352) 92 07 31-1
Fax: (+352) 92 07 31-300
info@bmedicalsystems.com