Author: Nancy Castaneda, Account Analyst and Krystal Gaskill, Manager, Pharmacy Services
Mastering short-cycle rules can be challenging and confusing. Coding practices can negatively impact your reimbursements if not followed according to Centers for Medicare and Medicaid Services (CMS) short-cycling guidelines. To help avoid the headaches that may accompany common pitfalls, like under-reimbursements due to improper coding, it is important to identify the issues and have the knowledge to remediate them to maximize reimbursements and prevent audits. In this article, you will learn the basics of appropriate coding, how to identify exceptions to short-cycling rules, and how coding ensures transparent communication with the plan.
What is short-cycle prescription dispensing?
Short-cycling is the process of dispensing brand oral solids and New Drug Application (NDA) generic drugs in a 14-day or less supply to Medicare Part D plans (National Community Pharmacists Association, 2012).
What is the purpose of short cycling?
In 2013, CMS mandated the short-cycle rules to reduce prescription waste. CMS required many brand oral solid medications dispensed to skilled nursing facilities in 14-day supplies or less based on the industry standard length of stay for most patients. To make billing more transparent with the short-cycle rules in place, CMS defined several codes pharmacies must use telling why, when, and how a drug is being dispensed (Bloemke, 2017), (Medicare Part C, Medicare Part D, Prescription Drugs, Compliance, 2015).
What claims are required to be short-cycled?
Short-cycling is a requirement limiting any brand oral solid or NDA generic drug to a 14-day supply or less when dispensed to a skilled nursing facility (SNF), using a residence code of 03 and billed to a Medicare Part D plan. NDA generic drugs are considered generic, which would typically allow them to be billed as prescribed; however, because of their application to the Food and Drug Administration (FDA), these NDCs are required to be handled in the same manner as a brand oral solid and should be dispensed in a quantity of 14-days or less.
There are exceptions to this 14-day rule, which will be touched on in a later section of this article.
These codes assist the Medicare Part D Pharmacy Benefits Manager (PBM) in determining the rate at which a pharmacy will be reimbursed based on the patient, the pharmacy, and the facility in which patient resides (Berwick, 2011). Pharmacies must understand the definitions of these codes and apply them correctly to ensure the appropriate reimbursement on those claims.
Common Codes and Definitions:
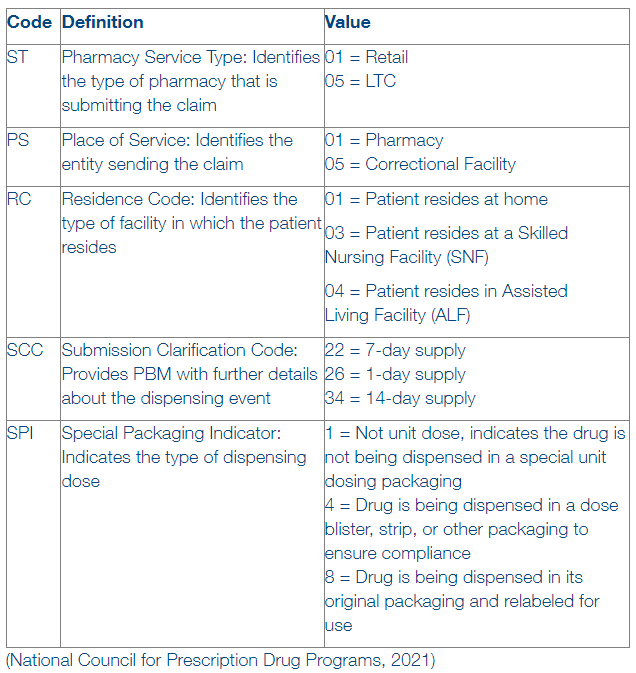
When these codes are applied inaccurately, lower than expected reimbursement may occur or a payer audit could be triggered. The example below should help explain the story these codes can provide.
Patient example: Mary
Mary was critically ill and was hospitalized for several days. As Mary's health improved, she was transitioned from a hospital environment to an SNF establishment. The facility has a contract with ABC Pharmacy to fill prescriptions for Mary. The doctor called in a 30-day supply of a medication to ABC Pharmacy for Mary. What should the pharmacy do?
The pharmacy should ask themselves the following questions:
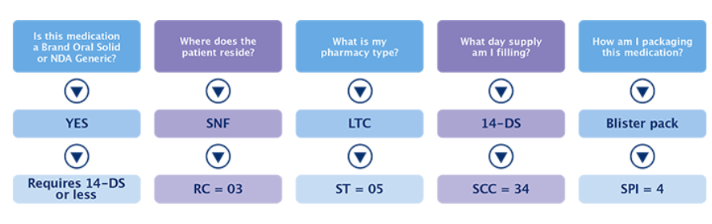
These are some of the questions that every biller should be asking themselves when processing prescriptions. The codes tell the PBM the story and create transparency related to the reimbursement rate the pharmacy yields. Using these codes accurately is critical for proper reimbursement.
Short-cycling exemptions
The most common reason a brand oral solid or NDA generic could be excluded from short-cycling is that the manufacturer has stated the packaging cannot be broken, and the drug requires dispensing in its original packaging. One example of this is Linzess; due to moisture concerns, Linzess cannot be opened before delivering it to the patient and excludes the medication from short-cycling rules. Another reason for exclusion could be a single course of an antibiotic. If the treatment is 15 days and is only for a single course (not repeating), often the full 15 days can be dispensed all at once.
Because exemptions are allowed within short-cycling, pharmacies must utilize the appropriate Submission Clarification codes (SCC) to indicate why a particular medication is dispensed for a day supply greater than 14. For retail pharmacies, SCCs are often used to allow for vacation overrides and lost or stolen medications. For LTC pharmacies, SCCs can help tell the PBM how many days of medication were delivered to the facility. Correct use of SCCs can help pharmacies receive appropriate reimbursements and avoid audits and underpayments.
How can we help?
Net-Rx provides an array of comprehensive tools to help combat short-cycling confusion. Script-IQ, a pre-edit solution, verifies short-cycle codes correlate based on the drug dispensed and facility type. If a discrepancy is identified, Script-IQ provides the pharmacy an opportunity to review the data in the claim for accuracy. Edit-Rx, a post-edit solution, includes various reporting to identify all claims that may require a second look. As an example, the SCC report, when used as a tool to proactively verify proper coding for dispensing practices, can help minimalize disruption when audits do occur. Additionally, your dedicated Net-Rx analyst can provide guidance in correcting errors and making business decisions.
If you would like to learn more about how Net-RxTM services and tools can you help your pharmacy, please chat with a live representative now or contact us here.
References
Berwick, D. M. (2011, March 16). Medicare Program; Changes to the Medicare Advantage and the Medicare Prescription Drug Benefit Programs for Contract Year 2012 and Other Changes. Retrieved from Federal Register: https://www.federalregister.gov/documents/2011/04/15/2011-8274/medicare-program-changes-to-the-medicare-advantage-and-the-medicare-prescription-drug-benefit#h-36
Bloemke, R. (2017, May/June). Navigating Short-Cycle Billing. Retrieved from Computertalk: https://www.computertalk.com/navigating-short-cycle-billing/
Medicare Part C, Medicare Part D, Prescription Drugs, Compliance. (2015, February 06). CMS finalizes program changes for Medicare Advantage and Prescription Drug Benefit Programs for Contract Year 2016. Retrieved from cms.gov: https://www.cms.gov/newsroom/fact-sheets/cms-finalizes-program-changes-medicare-advantage-and-prescription-drug-benefit-programs-contract
National Community Pharmacists Association. (2012, December). ncpa.co. Retrieved from Overview of Short-Cycle Dispensing for Long-Term Care: http://www.ncpa.co/pdf/leg/dec12/overview_short_cycle_dispensing.pdf
National Council for Prescription Drug Programs. (2021, May). NCPDP WG9 Medicare Part D Questions and Answers. Retrieved from ncpdp.org: https://www.ncpdp.org/ncpdp/media/pdf/ncpdp_wg9_medicare_partd_faq.pdf
This post is related to:
Finance: Worker's Comp, Claims, PA Management, Adjudication