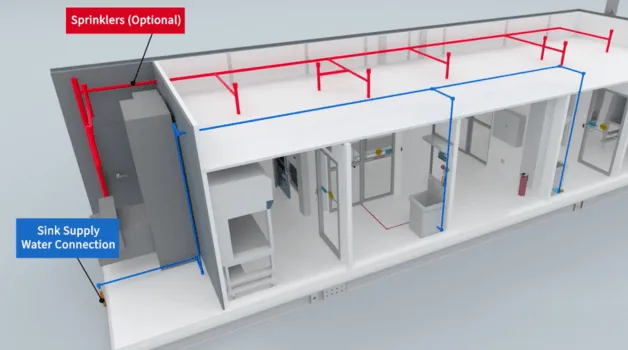
Healthcare facilities that perform sterile or hazardous drug compounding must clearly understand the difference between USP <797> and USP <800> and how each regulation impacts cleanroom design. While both standards regulate compounding pharmacy environments, they serve different purposes and require different cleanroom strategies.
For hospitals, health systems, oncology centers, and outsourcing facilities, understanding USP <797> and USP <800> requirements is critical before building, renovating, or deploying a cleanroom solution, especially when evaluating permanent versus mobile cleanroom options. Read More >