Learn more about Highlighted Practice Settings insights and Pharmacy500 companies behind the industry in our Pharmacy500 Platinum Pages 2026 publication!
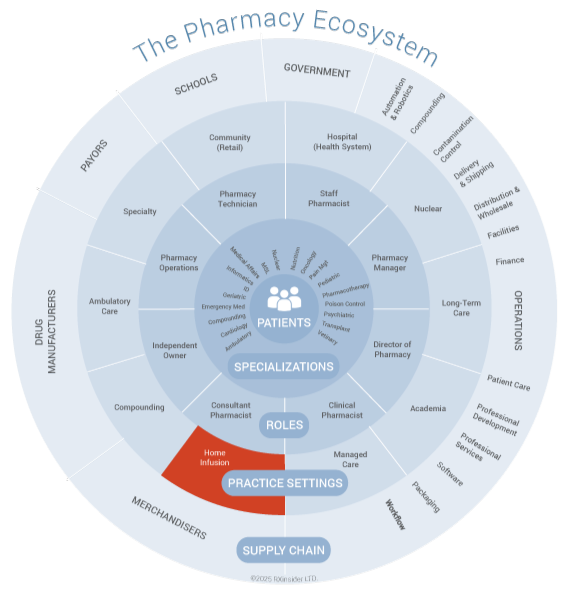
An infusion compounding pharmacy is a specialized pharmacy that prepares customized sterile medications for administration via infusion therapy, including intravenous (IV), intramuscular (IM), subcutaneous (SC), and epidural routes. These pharmacies operate under stringent standards for sterility and accuracy to meet the complex needs of patients requiring personalized infusion solutions for acute, chronic, or palliative care. Infusion compounding pharmacies are integral to delivering life-sustaining therapies for patients who require specialized, sterile medications, often in home-based or clinical infusion settings, and are closed to the public (closed-door pharmacy).


Company Background & History
Coldkeepers is a pioneer and leader in providing innovative thermal shipping solutions tailored to address the most demanding cold-chain challenges. Our proprietary shipping products, combined with our adaptable, logistics-based service, maintain and preserve the cold-chain process for the unique needs of clients in the pharmaceutical, health, and life science industries.
Co-founded in 1998 by Lang Flowers, III (CEO) and Lionel de Bazelaire (COO), and headquartered in picturesque Thomasville, Georgia, Coldkeepers remains equally committed to environmentally responsible packaging design, domestic production, and continuing innovation.
Product Overview
Coldkeepers’ patented shipping supplies provide state-of-the-art protection for your temperature-sensitive products from warehouse to doorstep. Ranging from curbside recyclable one-piece insulated box liners to thermally insulated mailers, pouches, and gel packs, our product line is designed to address the industry’s toughest cold-chain standards.
Coldkeepers products come with leading cold supply chain expertise and experience. Our trained specialists will work closely with your team to develop a tailored thermal packaging solution so your products can reach their destination safely.
Features & Options
Introducing the Penguin Mailer: Superior Cold-Chain Protection
At Coldkeepers, we’re proud to present our cutting-edge solution to cold-chain shipping — the Penguin Mailer. Engineered with superior craftsmanship and robust materials, the Penguin Mailer offers unparalleled protection for products in transit, setting a new industry standard. The Penguin Mailer ships flat at 15% of its normal size. This efficient packaging solution dramatically reduces storage space requirements and freight costs, making it ideal for small specialty pharmacy locations. Available in sizes: 9″x12″, 12″x16″, 15″x20″, and all with 1.375″ foam. The Penguin Mailer covers 95% of pharmacy shipments.
Curbside Recyclable One-Piece Insulated Liner
Our newest liners are suitable for shipments of controlled room temperature, refrigerated, or frozen pharmaceuticals between 2-8°C for 24 to 48+ hours (use with gel packs or dry ice to extend transit times). They boast a flexible design that allows them to ship flat and save on inbound freight and warehousing expense. Manufactured from postindustrial paper products, our liners are not only certified curbside recyclable by Western Michigan University but also third-party ISTA lab tested and certified by world renown WuXi AppTec testing lab.
One-Piece Extreme Liner
Third-party tested and certified by WuXi AppTec, these liners will keep your temperature-sensitive products at the right temperature for one to two day shipments. Our team of dedicated specialists can also help you design a custom pack-out to ensure your products arrive without breaking the cold chain. Our one-piece extreme liners can be used in conjunction with gel packs or dry ice for products that must be kept at a preset temperature for extended periods of time.
Extreme Thermal Mailers
Our Extreme Mailers are great for small parcels of temperature-sensitive medications when you need to ensure your prescription drug shipments stay at the right temperature during transit. Designed to fit inside UPS and FedEx Express boxes, the Extreme Insulated Mailers feature an open cell foam inner liner providing your products with unsurpassed levels of thermal and shock absorbing protection.
Insulated Pouches
Our line of Insulated Pouches features an easy-to-use double track pressure closure to ensure a tight and leakproof seal. They keep items cold for several hours or longer when paired with a frozen gel pack, ice or dry ice. Our exclusive line of durable pouches with built-in gel packs makes achieving those longer cooling times easy and encourages reuse. Simply freeze the pouch to charge the built-in gel pack, then you (or your customer) can reuse it over and over.
Experience the Coldkeepers Difference
More Information
To request a sample or connect with our experts, please visit our website www.coldkeepers.com or contact us at


Verity’s mission is to achieve pharmacy excellence through highly efficient, game-changing technology automation and exceptional support, enabling every customer to maximize their savings opportunity. It is built on a partnership rooted in trust and confidence in the work we do together. Founded in 2015 to support 340B participants, Verity now offers pharmacy purchase optimization solutions beyond 340B for all healthcare entities, that also meet the needs of our existing customers. We do this while staying true to our core focus of pharmacy supply chain solutions.
• Recognized as Best in KLAS: 340B Management Systems for seven years
• Featured in the 2024 KLAS Emerging Solutions Top 20 Report for VERISAVE® Purchase Optimization.
Contact Verity today to learn all the ways we can help support your mission through optimized pharmacy savings opportunities.

Simplifi 797 is turnkey pharmacy compliance software that automates operations required to comply with USP chapters 795, 797 and 800. The customizable interface eliminates the paper and binder clutter of the inspection process, reducing time and effort for your team. We design the expert content, task management, and alerts to ensure you’re operating safely and compliantly—and we make them available on a mobile-friendly electronic platform, so you can provide the inspector everything they need at the touch of a finger.
Unlike other products that stop at inspection readiness, Simplifi 797 helps you continuously improve your practice with customizable analytics, up to 45 hours of continuing education and regularly updated best practices from industry experts.
Backed by Wolters Kluwer’s 200 years of leading domain expertise and a team of internal compounding compliance experts, Simplifi 797 handles the ‘what and how’ of USP Chapters 795, 797 and 800, so you can optimize operations, maximize staff competency, and pass every inspection with confidence.
The solution is custom-developed so each component aligns with the next, ensuring that everything from policies and procedures to training and task management are perfectly fit to your needs.
Streamline cleanroom operations and enhance compliance tracking documentation with task management, environmental sampling, master formulation templates and compounding logs. Easily manage multiple facilities from one centralized location.
The continued USP <795> and <797> revision conversations are drawing closer to finalizing the chapters, and the enforcement timeline is looming. The experts at Simplifi 797 are preparing so customers will be compliant at enforcement—we are enhancing our functionality and content to support the changes and developing resource documents to help you navigate the differences.
State-of-the-art analytics provide compliance activities and trends specific to your compounding sites, Quality Scorecard reports and more, to ensure a trouble-free inspection.
Staff receive ACPE-approved web-based training through an exlusive parternship with CriticalPoint that is updated as regulations evolve. Access expert-written observational competencies, an easy-to-use scheduler and a customizable competency builder.
When you purchase Simplifi 797, you’re not just acquiring a quality management system. You’re gaining a full support and success team dedicated to helping you get the most out of your compliance solution. First, our implementation team works with you to assess your situation, ensuring the solution is tailored specifically to your needs, and on your timeline. Once set up, you’ll have a 24/7 support team for any ongoing assistance. Plus, your dedicated Customer Success Manager is committed to facilitating your compliance success.
Finally, the application and its expert content are updated quarterly based on customer feedback and industry trends, so you have access to the latest best practices. Wolters Kluwer’s Simplifi 797 makes it easy to be compliant.


For over 40 years, NuAire has consistently led the pharmacy compounding industry in safety standards and customer satisfac¬tion. We obsess over our customers’ needs and tailor unique solutions to meet those needs. With invaluable input from customers like you, we now offer a wide range of laminar airflow workstations (LAFWs), containment ventilated enclosures (CVEs), biological safety cabinets (BSCs), ultralow temperature (ULT) freez¬ers, Restricted Access Barrier Systems (RABS), and much more. With our cutting-edge robotic sheet metal facility, we can deliver you custom, innovative pharmacy compounding solutions in addition to the standard products that we sell.
The LabGard Class II Type A2 & Type B2 Biological Safety Cabinets (BSCs) effectively operate as Containment-Primary Engineering Controls (C-PECs) for sterile hazardous pharmacy drug compounding. C-PECs provide USP 800 compliant product, personnel, and environmental protection that can be externally exhausted after passing through HEPA filtration.
The PharmaGard Restricted Access Barrier System (RABS) series offers a physical barrier between personnel and product. The positive pressure Compounding Aseptic Isolator (CAI) protects the non-hazardous compounded sterile preparation inside, while our negative pressure Compounding Aseptic Containment Isolators (CACIs) offer containment for hazardous sterile preparations and a choice of a model that recirculates most air to the work zone or another that is total exhaust. NuAire offers accessories so that either of these CACI models can be connected to external exhaust in compliance with USP 800.
The AireGard Laminar Airflow Workstation (LAFW) series operates as a Primary Engineering Control (PEC) that complies with USP 797 by providing product protection by flushing the work surface with vertical or horizontal ISO class 5 air.
The LabGard NU-813 Containment Ventilated Enclosure (CVE) or Class I BSC also effectively operates as a C-PEC that complies with USP 800 for non-sterile hazardous drug compounding. The NU-813 offers personnel and environmental protection by creating a constant 80 fpm (0.41 m/s) air inflow that is exhausted through HEPA filters.
The Blizzard -86°C Ultralow Temperature (ULT) Freezer series provides consistent storage temperatures of -40°C to -86°C, ideal for vaccine storage, and are equipped with energy efficient compressor motors. All NuAire freezer models utilize environmentally friendly hydrocarbon (HC) refrigerants and vacuum insulated polyurethane (VIP) chamber walls. Available capacities range from 3.5 to 14.8 ft3 (100 to 828 L).
NuAire pharmacy compounding workstations can be fitted with an optional IV bar with 6+ SST hooks, mechanical auto-rising base stands, ergonomic arm attachments for computer monitor + keyboard/mouse, and glass sidewalls for added visibility.
The Pharmacy500 is awarded to 500 pharmacy supply chain businesses and associations that significantly impacted dispensing pharmacies in the U.S. over the past year.
The following are complimentary benefits extended to Pharmacy500 companies and associations (coming this spring!)


Personal protective equipment is the first line of defense against exposure to Hazardous Drugs and the most cost effective means to protect employees. Prudential Cleanroom Services’ garments also protect the product from the employees. Humans are one of the largest contamination contributors to any cleanroom environment unless they are gowned correctly.
CLEANROOM GARMENT SYSTEM FEATURES
PCS VALIDATED REUSABLE MOP PROGRAM
Why purchase mop heads and dispose of them when you can reuse them?
All Prudential Cleanroom Services products are Validated SAL 10-6 for our Aseptic customers.
Our mission is to become the nation’s most trusted independent specialty pharmacy consulting group, creating value for our clients and staff while catalyzing clinical, operational, and financial performance for our clients.
Partner with a consulting team that understands the history and trajectory of this marketplace while providing all of the tools and necessary guidance to guide your pharmacy operation towards leadership and success in the space. Sphinx vows to provide double and triple-digit ROI’s to our clients while providing bleeding-edge expertise and disruptive approaches towards success for our staff and partners. Find out more about our brands, approach, and solutions throughout our website, and feel free to reach out for further questions at our contact info below.
The Sphinx team is pleased to introduce our analytics and informatics division – SYLTOS – a splinter group of the Sphinx team focused on generating disruptive technologies and developing informatics verticals for our pharmacies, infusion centers, hospitals and provider’s offices.
SPRYNG Specialty Pharmacy Partner Network is a select group of accredited independent specialty pharmacies
Key insights from across the industry including pipeline reports, tools, and tactics to build out your operations. monitor your targets and hit your goals.


















Learn more about Highlighted Practice Settings insights and Pharmacy500 companies behind the industry in our Pharmacy500 Platinum Pages 2026 publication!
The 2026 Pharmacy500 Companies Market Teardown report provides a concise snapshot of the 2026 companies, highlighting new additions, examining regional distributions, practice settings, supply chain categories, and key conferences attended.
RXinsider’s Pharmacy500 Careers Outlook report identifies key employment trends and workforce dynamics across Pharmacy500 companies. Check out this report to learn more about open position trends and the current state of the job market impacting the pharmacy supply chain!
Fortune Magazine’s annual Fortune 500 ranks the largest U.S. companies by revenue. Many leaders in healthcare and pharmaceuticals drive medication production, distribution, and patient access. This report highlights their impact on the pharmacy industry.
Visante delivers an integrated specialty pharmacy and infusion solution designed to support specialty pharmacy development, infusion services, 340B optimization, medication access, and revenue cycle improvement. To learn more, visit their profile in the 20Ways Winter Hospital publication.
In this case study, we take a look at Leiters Health partnered with Mobile Infirmary to pilot ready-to-dilute concentrated norepinephrine vials, significantly reducing preparation and administration time and freeing up IV-room resources.
A pharmacy conference is a professional gathering designed for pharmacists, pharmacy technicians, and other healthcare professionals to exchange knowledge, discuss emerging trends, and advance the practice of pharmacy. The Pharmacy500 Conferences Report provides insight into the conferences, trade shows, and meetings organized by Pharmacy500 companies, featuring event comparisons, attendee breakdowns, and more.
RXinsider’s Pharmacy500 Academy now offers training on infusion pharmacy resources and breakdowns on companies that service the infusion market. Visit the Pharmacy500 Academy to learn more.
September 2, 2025 – Amneal Pharmaceuticals, Inc. (NASDAQ: AMRX) announced that it has begun U.S. commercialization of TEPADINA® (thiotepa) for Injection 200 mg in a patented multichambered bag…